Methane hydrate is a crystalline compound made from space-filling cages of water molecules that accommodate methane. It exists in much of the sediments that cover the oceanic floor as well as in permafrost. Research on methane hydrate nucleation kinetics is important for hydrate recovery and storage, developing kinetic hydrate inhibitors, and designing oil-gas transportation pipelines. Post-Doctor ZHANG Zhengcai, and his supervisor Prof. GUO Guangjun, Institute of Geology and Geophysics, Chinese Academy of Sciences(IGGCAS), together with their co-workers, have undertake molecular dynamics simulations to investigate the nucleation kinetics of methane hydrate.
Molecular dynamics (MD) simulations, which can provide detailed information at a molecular level, have become a powerful technique for investigating microscopic structures and dynamics present during the gas hydrate nucleation process. The macroscopic physical properties of the simulation system in MD can be derived from the averages of the microscopic properties of all microstates. The ensemble is a probability distribution over the microstates, its properties are controlled by three parameters among number of particles N, volume V, energy E, pressure P, and temperature T. Previous studies have shown that different ensembles affect the hydrate nucleation process. The work by ZHANG et al. represents the first systematic study on the effects of different ensembles on gas hydrate nucleation kinetics.
They performed 20 simulations for each of the NPT, NVT, and NVE ensembles. The simulation trajectories were analyzed with using the mean first-passage time (MFPT) method (Fig. 1). The fitting results of MFPT show that for the three different ensembles, the hydrate nucleation rate is quantitatively ordered as follows: NPT > NVT > NVE, while the sequence of hydrate crystallinity is exactly reversed. Interestingly, the largest size of the critical nucleus appears in the NVT ensemble, rather than in the NVE ensemble (Fig. 2). This may be explained by the higher order of the critical nucleus in the NVE ensemble. This implies that future studies on the critical nucleus of a hydrate should consider not only its size, but also its structure (or the order degree) because both factors can affect the free energy of the critical nucleus, the ultimate factor that controls the nucleation phenomenon of hydrates.
This study was published in the RSC journal of PCCP(Physical Chemistry Chemical Physics) in May 2016 (Zhengcai Zhang, Chan-Juan Liu, Matthew R. Walsh, Guang-Jun Guo. Effects of ensembles on methane hydrate nucleation kinetics. Physical Chemistry Chemical Physics, 2016, 18: 15602-15608).
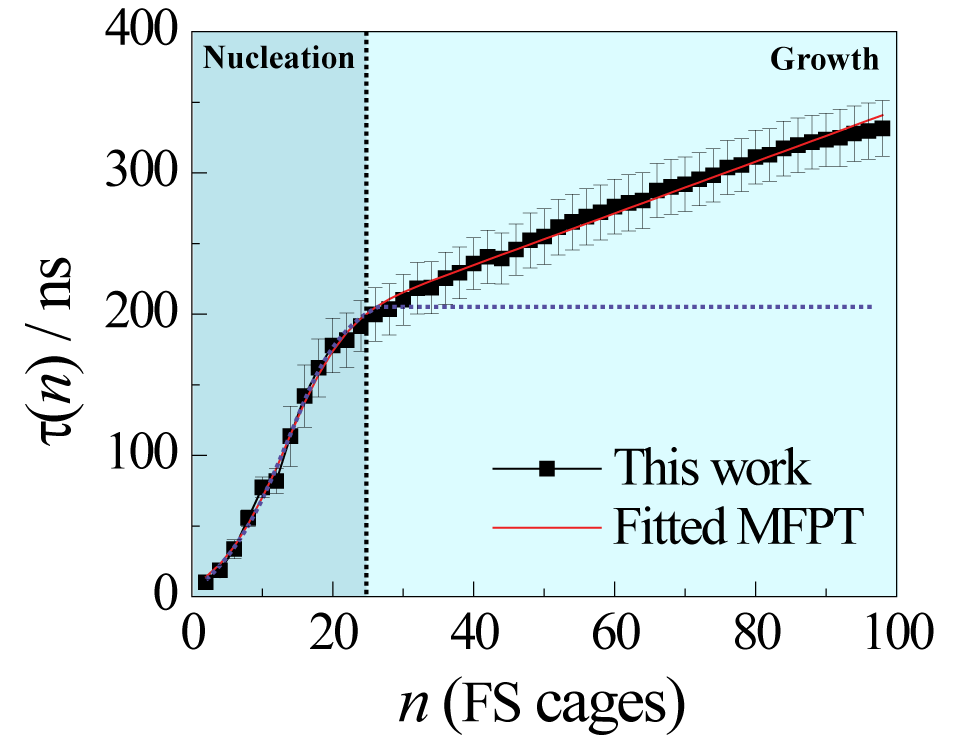
Fig. 1 Mean first-passage time as a function of cluster size (number of the FS cages) for the NPT ensemble.(Image by ZHANG et al.)
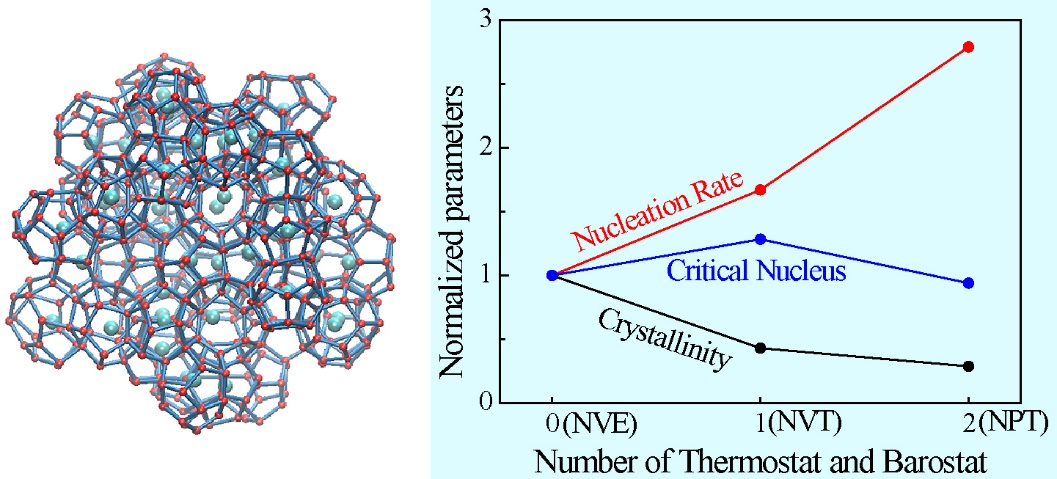
Fig. 2 The trends of the nucleation rate, critical nucleus, and crystallinity for the NPT, NVT, and NVE ensembles.(Image by ZHANG et al.)
Contact:
Prof. GUO Guangjun
INSTITUTE OF GEOLOGY AND GEOPHYSICS, CHINESE ACADEMY OF SCIENCES
Phone: 86-10-82998369
E-mail: guogj@mail.igcas.ac.cn